Gujarat firm Gets Notice For Contamination Probe After Eye Infections Reported By Sri Lanka
Gujarat-based Indiana ophthalmics has come under fire for allegedly distributing substandard eye drops in Sri Lanka. The Sri Lankan government lodged a complaint with the Indian government, claiming that more than 30 individuals have suffered eye infections as a result of using the company's eye drops.
In response to the allegations, India's leading pharmaceuticals export council, Pharmexcil, served a show cause notice to Indiana Ophthalmics on June 1, requesting an explanation regarding an internal investigation within the next two days.
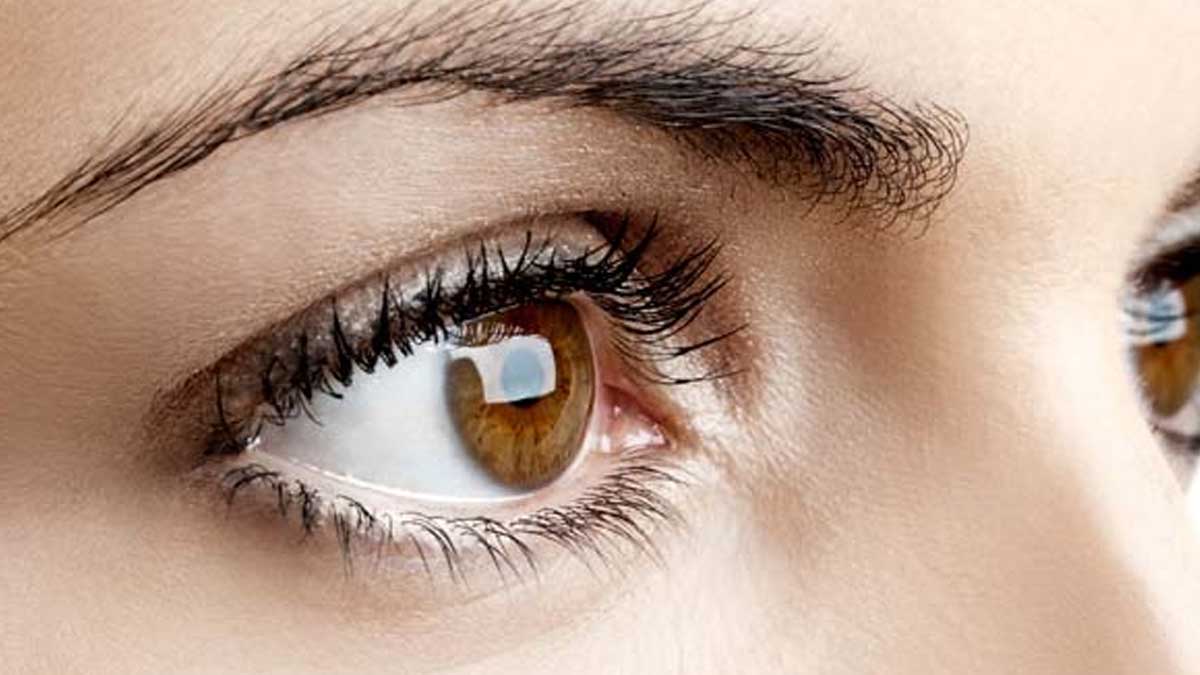
The Central Drugs Standard Control Organisation (CDSCO) has also initiated an inquiry into concerns surrounding the quality of Methylprednisolone eye drops manufactured by Indiana Ophthalmics.
In a letter written by Udaya Bhaskar, director general of Pharmexcil, expressed concern over the alleged supply of contaminated eye drops, emphasizing the detrimental impact on the reputation of the Indian pharmaceutical industry. Bhaskar further noted that such incidents could undermine the trust of international agencies in India's pharmaceutical exports.
"Upon failure of submission of requested information by 3 June 2023, your registration cum membership certificate (RCMC) will be suspended without any further notice," the letter read.
Indiana Ophthalmics has refuted the allegations of quality issues in its supplied eye drops.
This marks the fourth instance within a year in which drugs manufactured in India have been deemed contaminated in foreign countries.
Since October 2022, various importers of Indian medicines, particularly cough syrups, such as Uzbekistan, the Gambia, Marshall Islands, and Micronesia, have made allegations of contamination. These syrups, manufactured in India, were found to contain harmful substances like diethylene glycol (DEG) and ethylene glycol (EG).
In February, the US Food and Drug Administration issued a warning against using an Indian-made eyedrop, which was associated with an outbreak of drug-resistant bacteria.














 Click it and Unblock the Notifications
Click it and Unblock the Notifications