COVID-19 Vaccination Drive: 447 adverse events reported so far, 3 hospitalised
New Delhi, Jan 17: The central government on Sunday said that 447 cases of AEFI (Adverse Event Following Immunisation) were reported following the launch of massive nationwide vaccination drive.
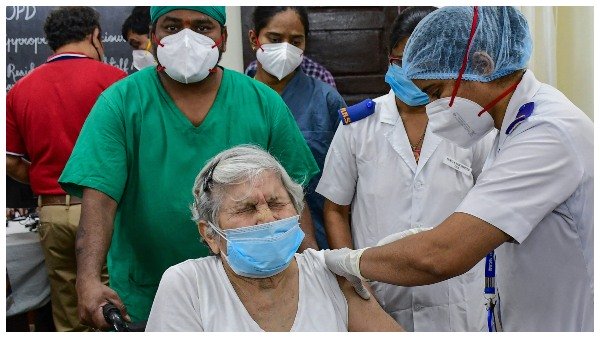
"A total of 447 adverse events following immunisation (AEFI) reported so far, of which only three cases required hospitalisation," Manohar Agnani, a senior health ministry official said.
While one of the three is still under observation in AIIMS Rishikesh, two were discharged from Northern Railway Hospital Delhi and AIIMS Delhi.
Agnani said that most of the adverse events following immunisation (AEFI), which were reported, were minor cases such as fever, headache and nausea.
"Today being Sunday, only six states conducted vaccination drive and in 553 sessions a total of 17,072 beneficiaries were vaccinated," he said.
The six states where the vaccination drive was carried out on Sunday are Andhra Pradesh, Arunachal Pradesh, Karnataka, Kerala, Manipur and Tamil Nadu, he added.
Prime Minister Narendra Modi rolled out the world''s largest inoculation drive against COVID-19 on Saturday and said the two vaccines being deployed will ensure a "decisive victory" for India against the pandemic.
India has approved two vaccines -- Covaxin developed by Bharat Biotech and Covishield from the Oxford/AstraZeneca stable being manufactured by the Serum Institute of India -- for emergency use in the country.
According to the government, the shots will be offered first to an estimated one crore healthcare workers and around two crore frontline workers, and then to persons above 50 years of age, followed by persons younger than 50 years of age with associated comorbidities.













 Click it and Unblock the Notifications
Click it and Unblock the Notifications