India Launches Home-Grown RT-PCR Kit For Monkeypox Detection
The World Health Organization (WHO) has declared a second public health emergency of international concern due to Monkeypox. The newly identified strain of the virus, Clad-1, is noted for its increased transmissibility and higher mortality rates, ANI reported.
In response to this urgent situation, India has developed an indigenous RT-PCR testing kit to combat Monkeypox, which has now been approved by the Central Drugs Standard Control Organisation (CDSCO). The IMDX Monkeypox Detection RT-PCR Assay, produced by Siemens Healthineers, represents a significant advancement in the country's "Make in India" initiative and plays a crucial role in addressing the Monkeypox public health crisis.
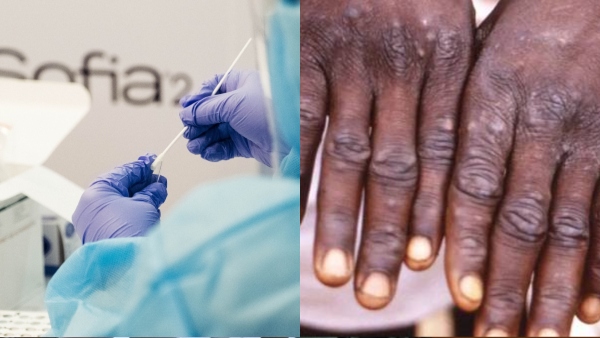
Siemens Healthcare Private Ltd announced that the IMDX Monkeypox Detection RT-PCR Assay will be manufactured at their molecular diagnostics unit in Vadodara, with an annual production capacity of 1 million reactions. The company is poised to make these kits widely available.
This cutting-edge assay targets two distinct regions of the viral genome, covering both Clade I and Clade II variants of Monkeypox. It offers comprehensive detection across different viral strains and integrates seamlessly into existing laboratory workflows with standard PCR setups, thereby eliminating the need for new equipment. The assay's compatibility with current COVID-19 testing infrastructure is expected to enhance efficiency.
Hariharan Subramanian, Managing Director of Siemens Healthcare Private Limited, emphasized the critical need for accurate diagnostics. "Providing India with advanced assay kits designed to detect Monkeypox is a proactive step in our battle against the disease. These kits underscore our commitment to improving 'Access to Care' and are a vital tool in saving lives through prompt and precise detection," he stated.
The test results from the IMDX Monkeypox Detection RT-PCR Assay will be available in just 40 minutes, significantly quicker than traditional methods, which typically take 1-2 hours. Clinically validated by ICMR-National Institute of Virology, Pune, the assay boasts 100% sensitivity and specificity. It adheres to Indian regulatory standards and meets the highest global benchmarks.















 Click it and Unblock the Notifications
Click it and Unblock the Notifications