Govt gives clean-chit to India-made eye drop under scanner in US
The eye drops made in Chennai by Global Pharma Healthcare, linked to at least three deaths and blindness in some other patients in the United States, are found to be of "standard quality", reports said on Tuesday.
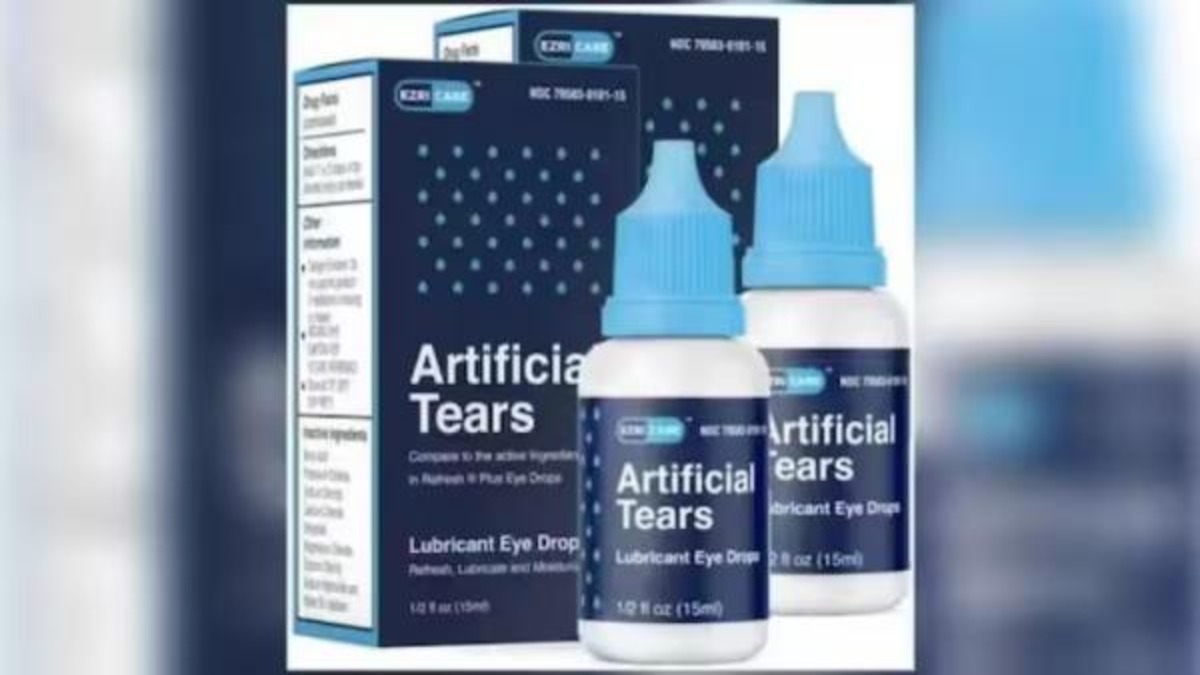
The test result of the samples, taken during a joint inspection by Tamil Nadu's Drug Controller and Central Drugs Standard Control Organisation in February, "came in favour of Global Healthcare Private Limited," NDTV reported quoting sources.
However, the pharmaceutical company was asked to "halt the production of the eye drops" to be on the safer side after top US medical watchdog raised concerns over it.
What did the US health regulators say?
The Centre for Disease Control and Prevention (CDC) has linked use of the eye drops to the likelihood of a highly drug-resistant bacteria gaining a foothold in the US. Citing the CDC, The New York Times earlier reported that three deaths, eight cases of blindness and dozens of infections had been traced to the eye drops with the brand name 'EzriCare Artificial Tears'.
Global Pharma recalls eye drops
Following this complaints, Global Pharma decided to recall 50,000 tubes of eye ointment in the US market due to bacterial contamination, according to the US Food and Drug Administration (USFDA).
As per its latest Enforcement Report, the US health regulator noted that the Chennai-based drug firm is recalling the affected lot of "Artificial Eye Ointment" from the US market. The lot has been manufactured by Chennai-based Global Pharma and distributed in the US market by New York-based Delsam Pharma, the USFDA said.
Stating the reason for recall, the US health regulator said: "FDA analysis found unopened tubes to be contaminated with bacteria." The company initiated the Class I recall on February 24 this year.
As per USFDA, a Class I recall is the most urgent of the 3 types of FDA recalls and usually pertains to defective products that can cause serious health problems. In a separate disclosure, USFDA stated that Mumbai-based Sun Pharma is recalling 1,920 bottles of Dofetilide capsules, which are used to treat irregular heartbeat.
The affected lot has been produced at Sun's Dadra-based plant, the USFDA stated. The US-based unit of the company - Sun Pharmaceutical Industries Inc - is recalling the lot due to "failed content uniformity specifications," it added.
The New Jersey-based firm initiated the Class III recall on March 9. As per USFDA, a Class III recall is initiated in a "situation in which use of, or exposure to, a violative product is not likely to cause adverse health consequences."














 Click it and Unblock the Notifications
Click it and Unblock the Notifications