After WHO nod, People vaccinated with Covaxin can now enter US from November 8: Details here
Washington, Nov 04: Travellers who have taken both doses of Bharat Biotech's Covid-19 vaccine Covaxin will be allowed to enter the US from November 8, new agency ANI reported. This comes a day after Covaxin gets emergency use approval from World Health Organisation (WHO).
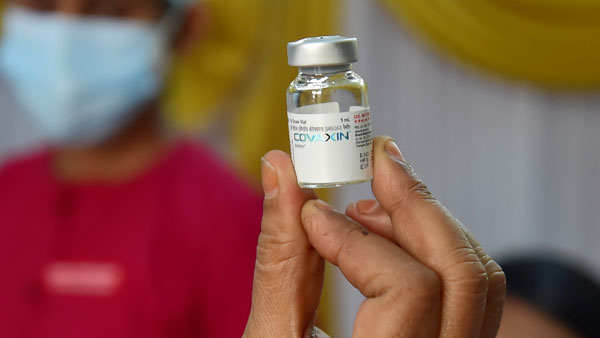
Talking to ANI over updating the approved travel list for Covaxin, CDC press officer Scott Pauley told ANI that US' travel guidelines apply to FDA approved on WHO Emergency Listed vaccines.
"CDC's travel guidance applies to FDA approved or authorized and WHO Emergency Use Listing vaccines and encompasses any new vaccines that may be added to either of those lists over time," he said.
The US Centre for Disease Control (CDC) yesterday acknowledged the WHO granting emergency use listing to Covaxin, developed by Bharat Biotech. The new travel rules will also accept travellers fully vaccinated with Pfizer-BioNTech, Johnson & Johnson, Moderna, Oxford-AstraZeneca, Covishield, Sinopharm and Sinovac.
Covaxin, India's indigenously developed coronavirus vaccine by Bharat Biotech, has been recommended for emergency use listing by the World Health Organisation (WHO). The Technical Advisory Group of WHO on Wednesday recommended Covaxin for emergency use status, according to PTI.
"Covaxin vaccine was also reviewed by WHO's Strategic Advisory Group of Experts on Immunization (SAGE), and recommended the use of this vaccine in two doses, with a dose interval of four weeks, in all age groups 18 and above," it said.
Following approval from WHO, Union Health Minister Mansukh Mandaviya thanked the world health body for Emergency Use Listing (EUL) to Made-in-India Covaxin.
Bharat Biotech was initially given permission for the sale and distribution of Covaxin with a shelf life of six months, which was later extended to nine months, a company spokesperson told PTI.
"The CDSCO has approved the extension of shelf life of Covaxin up to 12 months, from the date of manufacture. This approval of shelf life extension is based on the availability of additional stability data, which was submitted to CDSCO," Bharat Biotech said in a tweet.














 Click it and Unblock the Notifications
Click it and Unblock the Notifications