Indian-made syrups linked to child deaths in Gambia: US body report
A joint probe by the Centers for Disease Control and Prevention (CDC) of the US and the Gambian health authorities has suggested a strong link between the death of many children in Gambia and the consumption of made-in-India cough syrups that were allegedly contaminated. In October, the World Health Organization (WHO) had issued an alert stating that the four cough syrups being supplied to Gambia by the India-based Maiden Pharmaceuticals Ltd were of substandard quality and claimed that they were linked to the death of many children in Gambia.
A CDC report released on Friday stated, "This investigation strongly suggests that medications contaminated with Diethylene Glycol [DEG] or Ethylene Glycol [EG] imported into the Gambia led to this Acute Kidney Injury (AKI) cluster among children." "Patients with DEG poisoning can experience a range of signs and symptoms, including altered mental status, headache, and gastrointestinal symptoms; however, the most consistent manifestation is AKI, characterized by oliguria (low urine output) or anuria, progressing over 1-3 days to renal failure (indicated by elevated serum creatinine and blood urea nitrogen)," read the report.
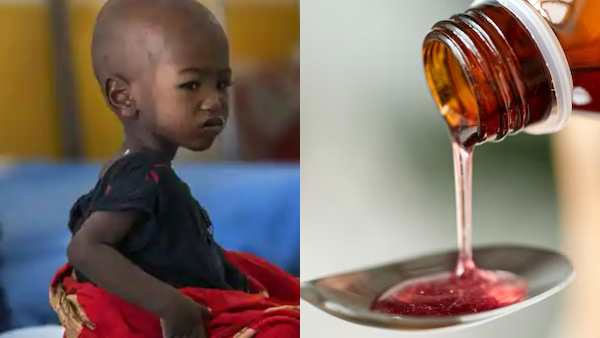
According to the CDC, they were contacted by Gambia's Ministry of Health (MoH) to assist in characterizing the illness (multiple cases of Acute Kidney Injury and deaths in children), describing the epidemiology, and identifying potential causal factors and their sources in August last year.
The report also said that in past DEG outbreaks, manufacturers have been suspected of substituting DEG in the place of more expensive, pharmaceutical-grade solvents.
"Among reports of AKI associated with DEG-contaminated medical products, this is the first in which DEG-contaminated medications were imported into a country, rather than being domestically manufactured," it said.
It further said medications for export might be subject to less rigorous regulatory standards than those for domestic use.
"Simultaneously, low-resource countries might not have the human and financial resources to monitor and test imported drugs," it stated.
Union Minister of State for Health Bharati Pravin Pawar in a reply to Lok Sabha on February 3 had said that after testing, the samples of the cough syrups have been declared to be of standard quality.
The samples were found to be negative for both Diethylene Glycol (DEG) and Ethylene Glycol (EG), Pawar had said in a written reply to a question.














 Click it and Unblock the Notifications
Click it and Unblock the Notifications