Foreign vaccines like Moderna, Pfizer likely to reach India sooner than expected
New Delhi, June 02: In an attempt to fast track the import of vaccines approved by countries and WHO for emergency use, the companies will not need bridging trials in India the Drug Controller General of India has said.
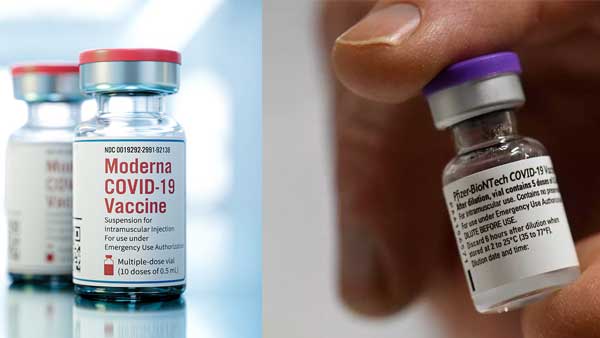
This move will clear the way for vaccines such as Pfizer and Moderna. They had requested the government for waivers such as post approval local trials and indemnity. The DGCI said that it has waived the requirements for these companies to carry out post launch bridging trials.
In a letter chief of DGCI V G Somani said that the decision was taken due to the huge requirement for vaccinations in India. The same was approved by the National Expert Group on Vaccine Administration for COVID-19.

"It has been decided that for approval of COVID-19 Vaccines in India for restricted use in emergency situation which are already approved for restricted use by US FDA, EMA, UK MHRA, PMDA Japan or which are listed in WHO Emergency Use Listing (EUL) and which are well established vaccines from the stand point that millions of individuals have already been vaccinated with the said vaccines, the requirement of conducting post approval bridging clinical trials and the requirement of testing of every batch of the vaccine by the Central Drugs Laboratory (CDL), Kasauli can be exempted, if the vaccine batch/lot has been certified and released by National Control Laboratory of Country of Origin," the letter by Dr. Somani read.













 Click it and Unblock the Notifications
Click it and Unblock the Notifications