Govt to get 1 crore doses of Zydus Cadila vaccine by December
New Delhi, Nov 18: Zydus Cadila's COVID vaccine for persons above 18 years of age will be supplied to government by December. Health ministry has placed an initial order for one crore doses of the vaccine.
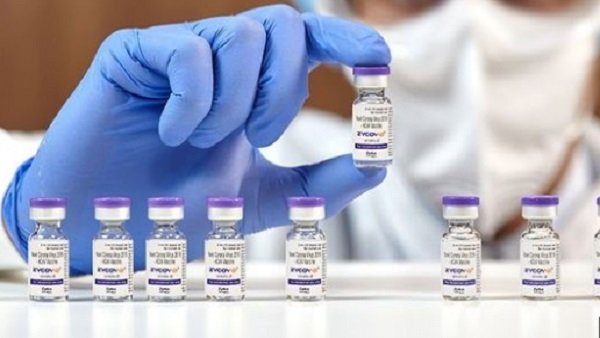
ZyCov-D, which has been cleared by India's drug regulator for those aged 12 years and above, will be given only to adults as of now under the national Covid vaccination drive.
Currently, three vaccines -- Covishield, Covaxin and Sputnik V -- being administered in the country are only for those above 18 years of age. All of them are two-dose vaccines.
Union Health Minister Mansukh Mandaviya on Thursday said the government does not want to make haste about administering Covid vaccines to children and any decision in this regard will be taken based on expert opinion.
He said children are not being inoculated against COVID-19 on a large scale anywhere in the world, though it has been initiated in some countries in a limited manner.
For administering ZyCov-D to adults, frontline workers and vaccinators will be provided a brief training for using the needle-free applicator in actual field settings.
At present, every citizen aged above 18 years is eligible for Covid vaccination.
Drug firm Zydus Cadila on November 8 said it had received an order to supply one crore doses of its Covid vaccine, ZyCoV-D, to the Indian government at Rs 265 per dose.
The vaccine will be administered using a needle-free applicator as opposed to the traditional syringes. The applicator is called "PharmaJet".
PharmaJet is a needle-free applicator to ensure painless intradermal vaccine delivery, which also leads to a significant reduction in any kind of major side effects.
ZyCoV-D is the first DNA plasmid vaccine in the world for human use, developed indigenously by the company against COVID-19, Zydus Cadila said.
The three doses of ZyCoV-D are to be administered 28 days apart. The vaccine was given emergency use authorisation (EUA) by the Indian drug regulator on August 20.















 Click it and Unblock the Notifications
Click it and Unblock the Notifications