Biggest tranche of Sputnik V vaccines lands in Hyderabad
New Delhi, June 01: The biggest tranche yet of Russia's Sputnik V vaccine of nearly 3 million doses landed in Telangana from Russia early on Tuesday.
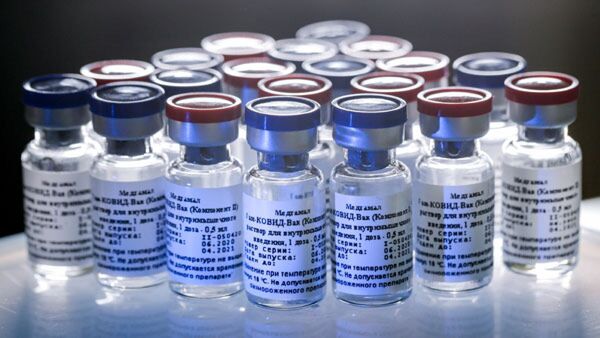
A GMR Hyderabad Air Cargo (GHAC) press release said the vaccine consignment arrived from Russia on a specially chartered freighter RU-9450 which touched down at Hyderabad Airport at 03.43 hours.
'While GHAC has already handled several import shipments of vaccine prior to this, todays shipment of 56.6 tonnes of vaccines is the single largest import shipment of COVID-19 vaccines handled in India till date.This shipment completed all processes and was dispatched in less than 90 minutes,' it said.
The Sputnik V vaccine requires specialised handling and storage, required to be kept at a temperature of -20 C.
Government had earlier said that six lakh imported double-dose of Sputnik-V will be available in May 2021, one crore imported doses in June 2021 and 2.8 crore doses (2.4 Crore imported and 40 Lakh manufactured in India) in July 2021.
August 2021 onwards, locally manufactured Sputnik V vaccine will be available in the domestic market and technology-transfer arrangement for manufacturing of Sputnik-V vaccine has been firmed up with six Indian manufacturers.
The two-dose Sputnik V had received the Indian drug regulator''s permission for restricted emergency use with certain conditions on April 12. Dr Reddy''s Laboratories is importing the vaccine for use in India.
Dr Reddy's Laboratories Ltd had applied for the grant of permission to import and market Gam-COVID-Vac combined vector vaccine, popularly called Sputnik-V, developed by Gamaleya Institute, Russia for Emergency Use Authorisation.
The Gam-COVID-Vac combined vector vaccine (Component I and Component II) has been developed by the National Research Center for Epidemiology and Microbiology of the Ministry of Health of the Russian Federation and is approved in 30 countries across the world.
DRL has collaborated with the Russian developer for obtaining regulatory approval for import for marketing in India.
The interim results of safety immunogenicity and efficacy from the Russian phase-3 clinical trial have been published in the Lancet journal.
With many states reporting shortage of vaccines, the government is trying to secure imported jabs, including of Russia''s Sputnik vaccines, while talks are also underway for COVID-19 vaccines developed by US majors Pfizer and Moderna.
Besides, the government is also looking to ramp up domestic manufacturing of vaccines, including those made by Serum Institute and Bharat Biotech, among other Indian manufacturers currently in various stages of their vaccine production.













 Click it and Unblock the Notifications
Click it and Unblock the Notifications