Bharat Biotech’s Covaxin phase-3 trials cross 13,000 volunteers
New Delhi, Dec 22: Bharat Biotech announced the successful recruitment of 13,000 volunteers and continued with the progress of achieving its goal of 26,000 participants for phase-3 clinical trial.
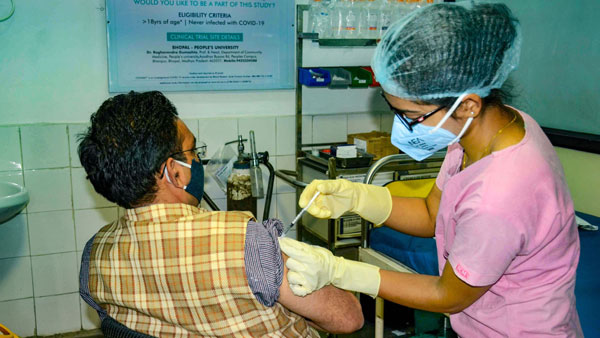
Phase-3 of the trials of Covaxin began in November. This is India's first and only Phase-3 efficacy study for a Covid-19 vaccine, and the largest phase -3 efficacy trial ever conducted for any vaccine in India. Covaxin has been evaluated in approximately 1,000 subjects in Phase I and Phase II clinical trials, with promising safety and immunogenicity results, with acceptance in international peer-reviewed scientific journals, the company said in a statement.
This is an unprecedented vaccine trial ever to take place in India, and we are overwhelmed with the steady rise in participation. We sincerely thank all the 13,000 volunteers across the country for their support in enabling us to bring out a safe and efficacious Indian vaccine for Covid-19. This pro-vaccine public health volunteerism is a morale booster for us to achieve our milestone target of 26,000 soon - Thank you volunteers. Thank you India," Suchitra Ella, joint managing director of Bharat Biotech, said.
In November Prime Minister, Narendra Modi visited the facility Hyderabad and interacted with the scientists. He also congratulated them for the progress they had made in the trials.















 Click it and Unblock the Notifications
Click it and Unblock the Notifications