Coronavirus: Bharat Biotech’s nasal vaccine gets approval for phase 2 clinical trial
New Delhi, Aug 14: The first nasal vaccine against COVID-19, developed by Bharat Biotech, has received regulator's nod for conducting phase 2 clinical trial, the Department of Biotechnology said on Friday.
Phase 1 clinical trial has been completed in age groups ranging from 18 to 60 years, it said.
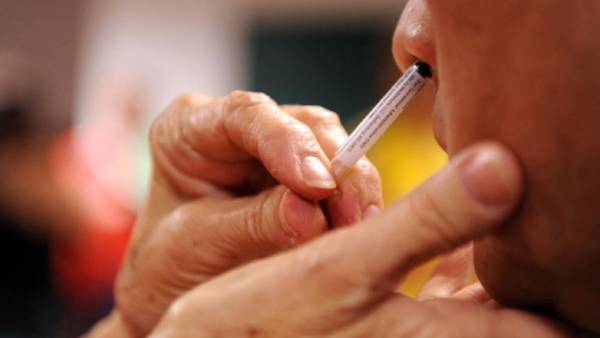
"Bharat Biotech's intranasal vaccine is the first nasal vaccine that has received the regulatory approval for phase 2 trials," the DBT said in a revised statement. In a statement issued earlier, the DBT had said the regulator's nod was given for phase 2 and 3 clinical trials.
The regulatory approval has been received for conducting "a Phase 2 randomised, multi-centric, Clinical Trial of Heterologus Prime-Boost Combination of SARS-CoV-2 Vaccines to evaluate the immunogenicity and safety of BBV152 (COVAXIN) with BBV154 (Adenoviral Intranasal COVID-19 vaccine) in healthy volunteers".
This is the first of its kind COVID-19 jab to undergo human clinical trials in India, it said. BBV154 is an intranasal replication-deficient chimpanzee adenovirus SARS-CoV-2 vectored vaccine.
"The company reports that the doses of the vaccine administered to healthy volunteers in the phase I clinical trial have been well tolerated. No serious adverse events reported," the DBT said. Previously, the vaccine was found to be safe, immunogenic and well tolerated in the pre-clinical toxicity studies. The vaccine was able to elicit high level of neutralising antibodies in animal studies, it added.
The Mission COVID Suraksha was launched to accelerate COVID-19 vaccine development efforts as part of the third stimulus package, Atmanirbhar 3.0. The focus of this mission is to consolidate and streamline available resources towards a warpath for accelerated vaccine development in order to bring a safe, efficacious, affordable and accessible COVID-19 vaccine to the citizens at the earliest.
"The department, through Mission COVID Suraksha, is committed to development of safe and efficacious COVID-19 vaccines. Bharat Biotech's BBV154 Covid vaccine is the first intranasal vaccine being developed in the country entering into late-stage clinical trials," Renu Swarup, Secretary, DBT and Chairperson, BIRAC said.














 Click it and Unblock the Notifications
Click it and Unblock the Notifications