
World Polio day: Why India can't afford to relax
New Delhi, Oct 24: India's success in eliminating wild polioviruses (WPVs) has been acclaimed globally. However, the detection of contaminated vials of type-2 polio vaccine among batches administered to children in some parts of the country has sparked concern in medical and government circles.
At least three batches of polio vaccines containing 1.5 lakh vials have been found contaminated with type 2 polio virus, putting at risk India's "polio free" status.
The polio virus causes paralysis medically known as an acute flaccid paralysis (AFP) - which is characterised by sudden muscle weakness, and fever in one or more limbs. AFP can occur due to many reasons, one of which is vaccine-linked.
Union Health Ministry has infact confirmed the contamination of few batches of vaccines of a particular manufacturer in three states: Maharashtra, Telengana and Uttar Pradesh.
However, all polio vaccine stocks of of that manufacturer have now been withdrawn.
The government has now urged all parents to get their children vaccinated against polio.

Virus and vaccine
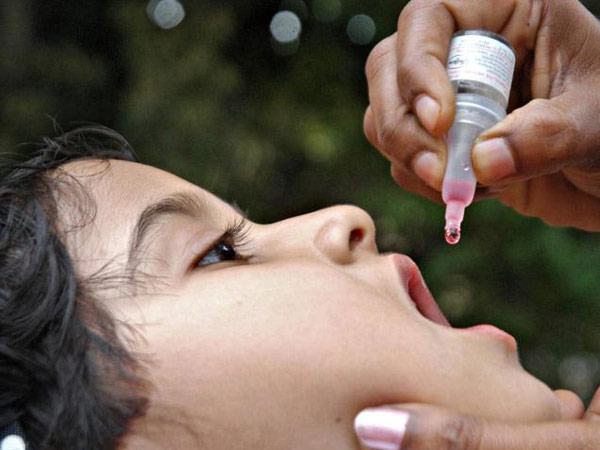
Polio, a permanently paralysing and sometimes fatal disease, can afflict children if they come into contact with any of the three distinct polio viruses-types 1, 2, and 3. So, from infancy up to six years of age, four doses of vaccination are administered, either orally or through an injection, to immunise them.
The injective method, known as inactivated poliovirus vaccine (IPV), contains only dead strains of the three types of the virus. The oral method, however, contains live, but less virulent, strains of the virus and can result in vaccine-induced polio. However, it is extremely rare-about one in 750,000 cases.
The vaccine virus is a problem because of all the oral vaccine strains, the strain of type-2 is most likely to cause vaccine-associated paralytic polio, which is why it was removed in the first place.

WHO downplays risk
The World Health Organisation (WHO) has said there was minimal risk of children contracting the polio virus in India from a tainted batch of vaccines. Shamila Sharma, a spokeswoman for the WHO, said any risk to children was "minimal" due to the high routine polio immunization coverage in India. The WHO funds and supports India's polio-control program.

Govt action
India's health ministry has said it is ramping up IPV vaccination "in the specified areas to reach out to such children who may have missed IPV".
"Type 2 polio vaccine virus traces, which have been found in bOPV vials, is the attenuated (weakened) polio virus and does not cause paralysis. It was also earlier used in tOPV till April 2016," a release by the Ministry of Health and Family Welfare said.
"The recipients of such vaccine will usually shed the vaccine virus through the fecal route for about 4-6 weeks after which it will die down," it added.
The government is looking to enhance immunity against type 2 polio virus further. Special mop up rounds for administering IPV are being conducted in the specified areas to reach out to such children who may have missed IPV.
India, often dubbed the pharmacy of the world, is home to thousands of factories churning out drugs and vaccines for sale globally, but local regulation is lax. In recent years, U.S. and European regulators have slammed many Indian drug factories for faulty manufacturing practices, often issuing warnings or bans.
India was officially declared "polio free" by the government in March 2014. The last case of wild polio virus type 2 in the country was in 1991.However, India continues to maintain a highly sensitive surveillance system for polio.


 Click it and Unblock the Notifications
Click it and Unblock the Notifications

































